 
They usually help in regulating and coordinating the products of an enzyme. They bind to the allosteric site of the enzyme changing the shape of the enzyme. They interfere with the enzyme that helped produce them. The lock-and-key model portrays an enzyme as conformationally rigid and able to bond only to substrates that exactly fit the active site. These inhibitors either bind to the active or allosteric site of an enzyme.įeedback inhibitors are the end products of reactions. 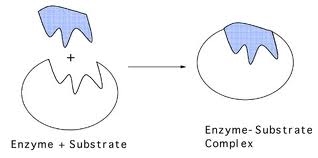
Irreversible inhibitors have two forms irreversible competitive inhibitors or irreversible noncompetitive inhibitors. Thus preventing the breakdown or formation of a molecule. When the inhibitor binds to the allosteric site, it causes a conformational shape change, preventing the enzyme's substrates from attaching to it. The matching between an enzyme's active site and the substrate isn’t just like two puzzle pieces fitting together (though scientists once thought it was, in an old model called the lock-and-key model). This area is known as the allosteric site. Rather they bind to a different area on the enzyme. Noncompetitive inhibitors, also known as allosteric inhibitors, do not compete with substrates for the active site. The lock and key model was first proposed by German scientist Emil Fischer in 1894. 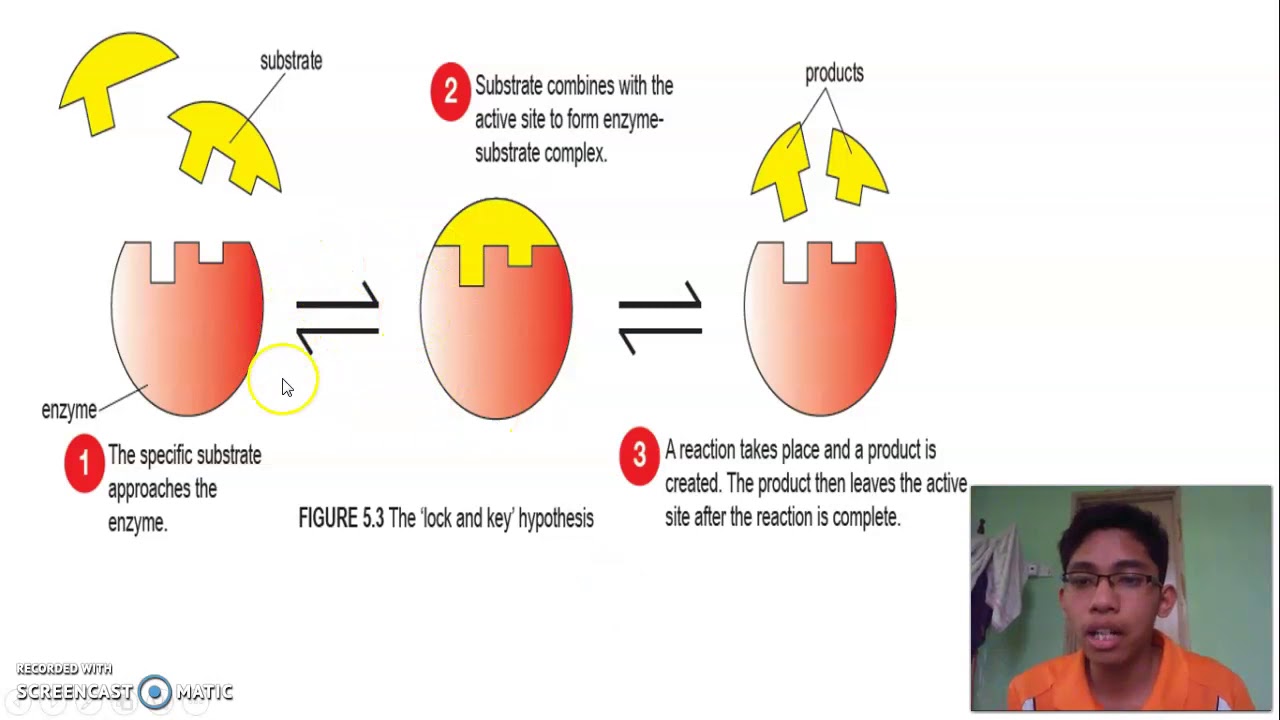
This specific binding allows the enzyme to lower the activation energy of the reaction, making it more likely to occur. lock-and-key mechanism A mechanism proposed in 1890 by Emil Fischer (18521919) to explain binding between the active site of an enzyme and a substrate. When they bind to the active site of the enzyme, they prevent the enzyme from breaking or creating molecules. The lock and key model of enzyme action proposes that the substrate fits into the active site of the enzyme like a key into a lock. Supported because enzymes are specific in the reactions they catalyse. A substrate will only fit the active site of one enzyme. Binding to enzymes brings reactants close to each other and aligns them properly, which has the same effect as increasing the concentration of the reacting compounds.There are four different kinds of inhibitors competitive inhibitors, noncompetitive inhibitors (allosteric inhibitors), irreversible inhibitors, and feedback inhibitors.Ĭompetitive inhibitors compete with the substrates of an enzyme at its active site. What is the lock and key model of enzyme action Proposes that enzymes work in the same way as a key operates a lock: each key has a specific shape that fits and operates a lock. The participating amino acids, which are usually widely separated in the primary sequence of the protein, are brought close together in the active site as a result of the folding and bending of the polypeptide chain or chains when the protein acquires its tertiary and quaternary structure. For many years, scientists thought that enzyme-substrate binding took place in a simple lock-and-key fashion. The lock-and-key model is used to describe the catalytic enzyme activity, based on the interaction between enzyme and substrate. The theory behind the Lock and Key model involves the complementarity between the shapes of the enzyme and the substrate. Amino acid side chains in or near the binding site can then act as acid or base catalysts, provide binding sites for the transfer of functional groups from one substrate to another or aid in the rearrangement of a substrate. The structural changes that occur when an enzyme and a substrate join together bring specific parts of a substrate into alignment with specific parts of the enzyme’s active site. (b) The enzyme conformation changes dramatically when the substrate binds to it, resulting in additional interactions between hexokinase and glucose. 
(a) The enzyme hexokinase without its substrate (glucose, shown in red) is bound to the active site. \): The Induced-Fit Model of Enzyme Action. The lock-and-key model portrays an enzyme as conformationally rigid and able to bond only to substrates that exactly fit the active site. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
